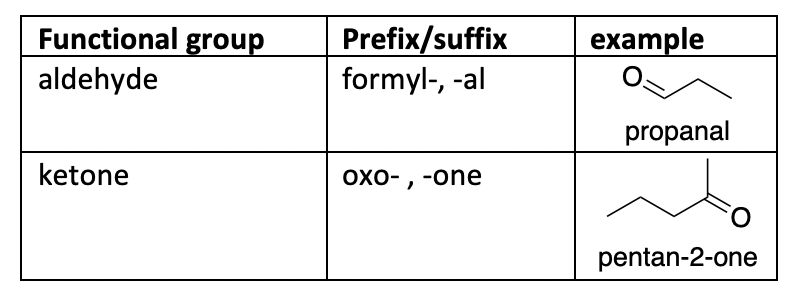
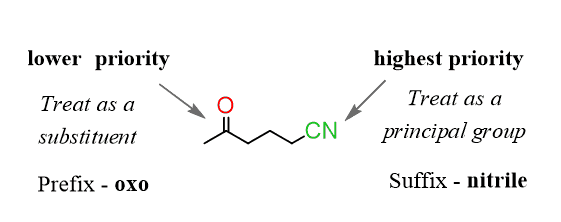
Novel Synthesis of 2-Oxo-3-butynoates by Copper-Catalyzed Cross-Coupling Reaction of Terminal Alkynes and Monooxalyl Chloride
How to know when to use keto and when to use oxo as both are prefixes for ketones in the nomenclature of polyfunctional organic compounds - Quora
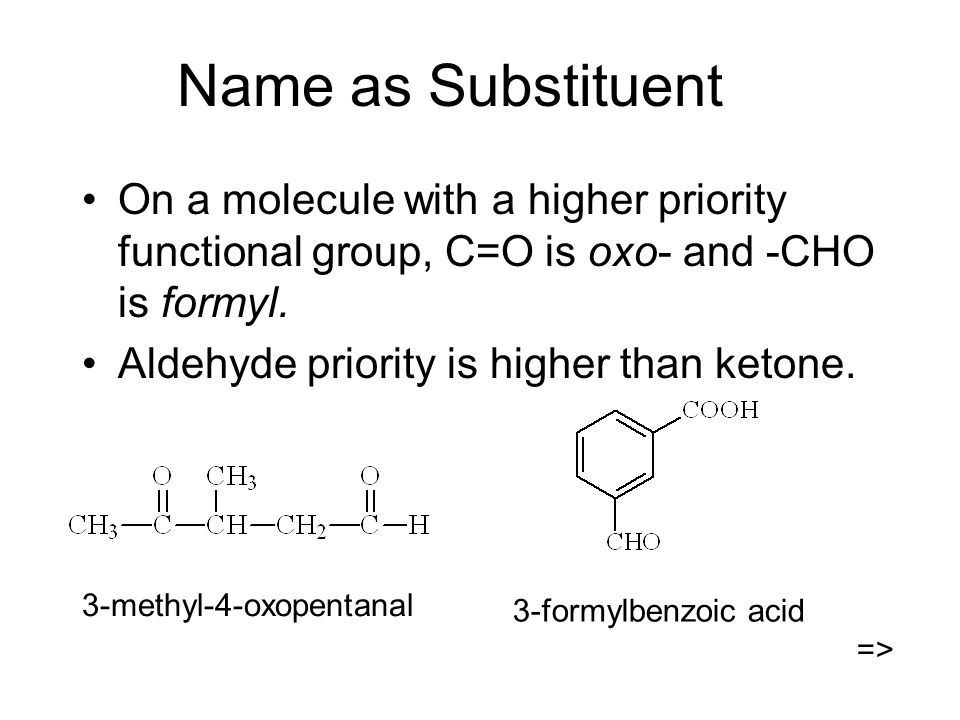
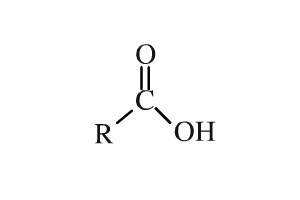
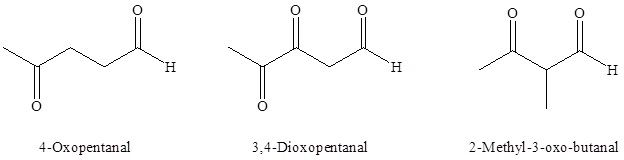
Name as Substituent On a molecule with a higher priority functional group, C=O is oxo- and -CHO is formyl. Aldehyde priority is higher than ketone. 3-methyl-4-oxopentanal. - ppt video online download