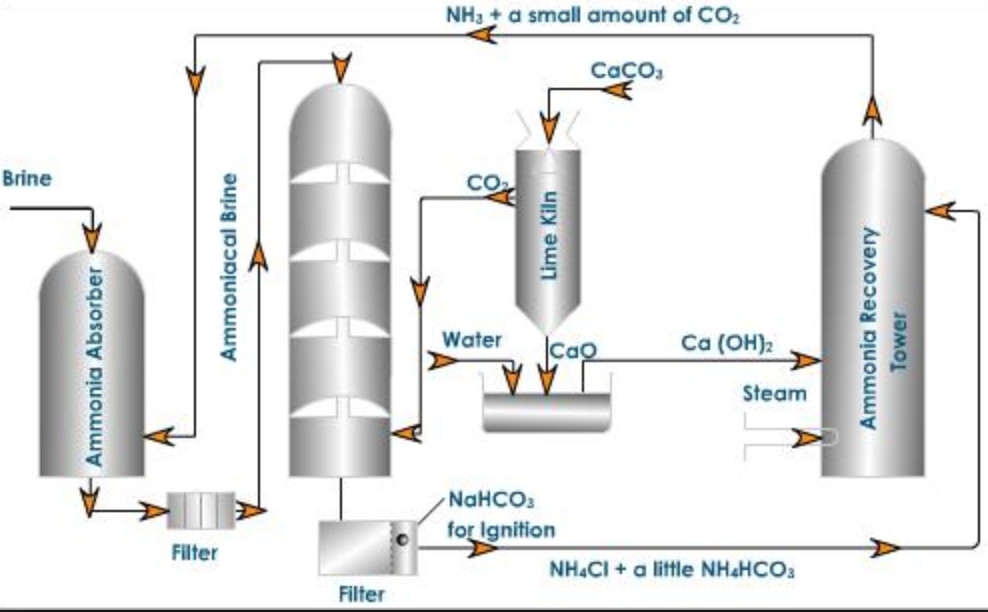
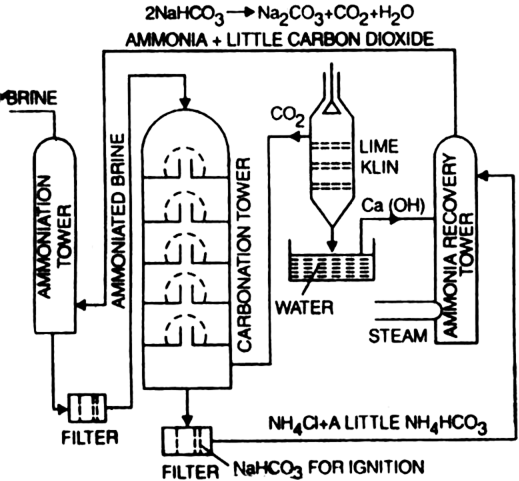
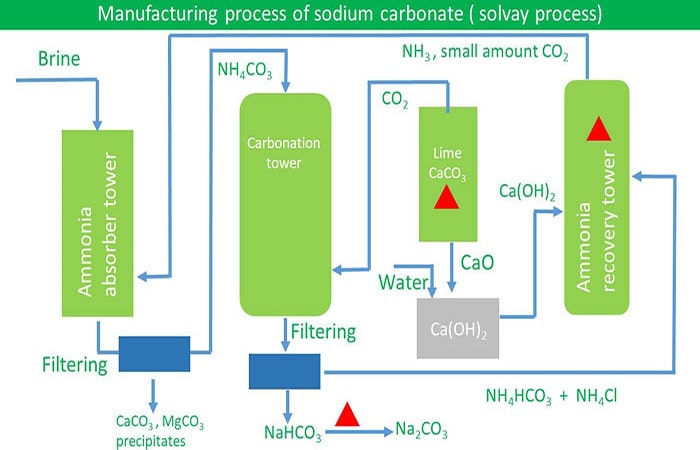
A modified Solvay process with low‐temperature calcination of NaHCO3 using monoethanolamine: Solubility determination and thermodynamic modeling - Wang - 2019 - AIChE Journal - Wiley Online Library

CHEManager International on Twitter: ".@SolvayGroup has announced plans to increase #soda ash capacity in #GreenRiverWY in order to meet global demand growth. It will also expand sodium bicarbonate output in #Devnya, #Bulgaria,

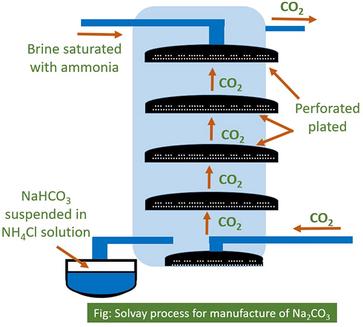



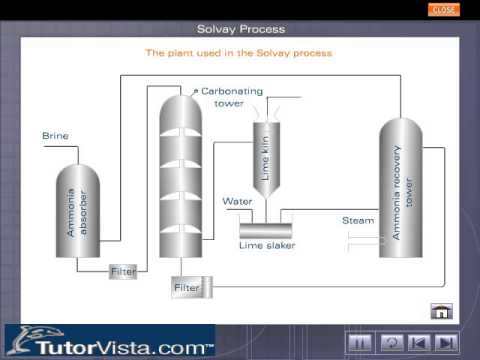
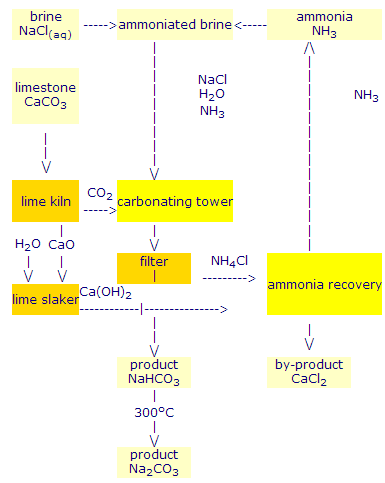

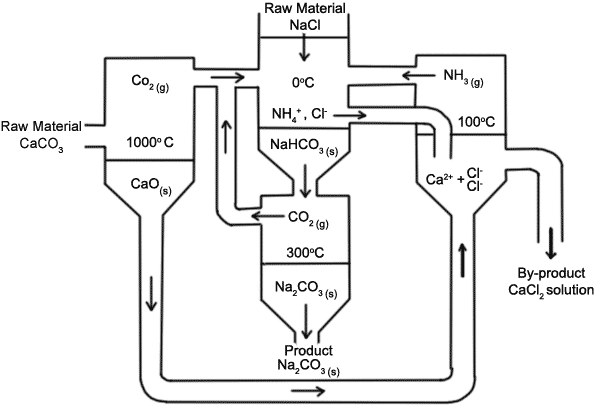
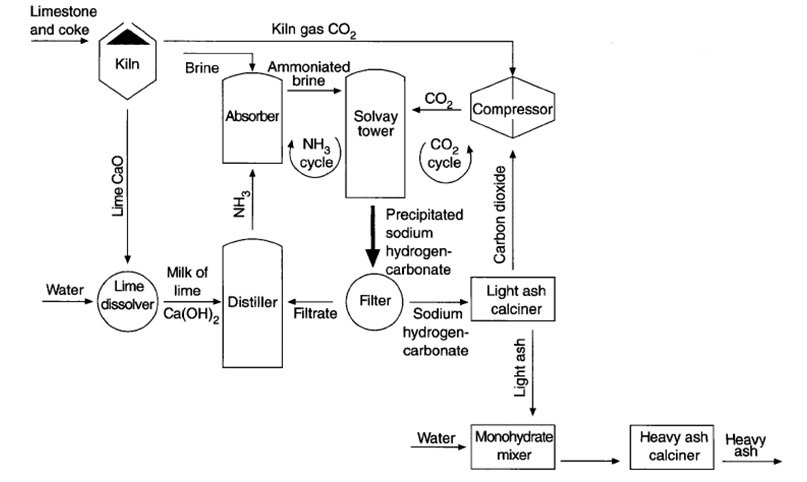






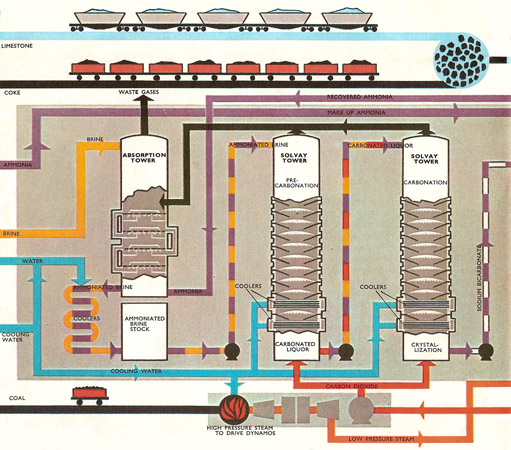
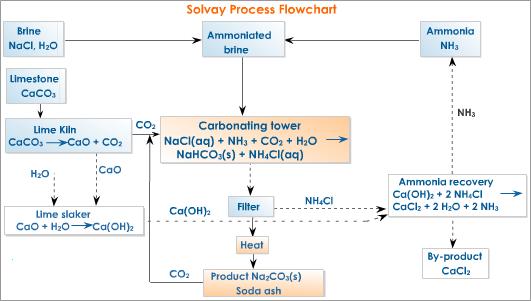
![PDF] The manufacture of soda ash in the Arabian Gulf | Semantic Scholar PDF] The manufacture of soda ash in the Arabian Gulf | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/12f9bf89433666383a43ba7191d119be5e6a6154/5-Figure1-1.png)