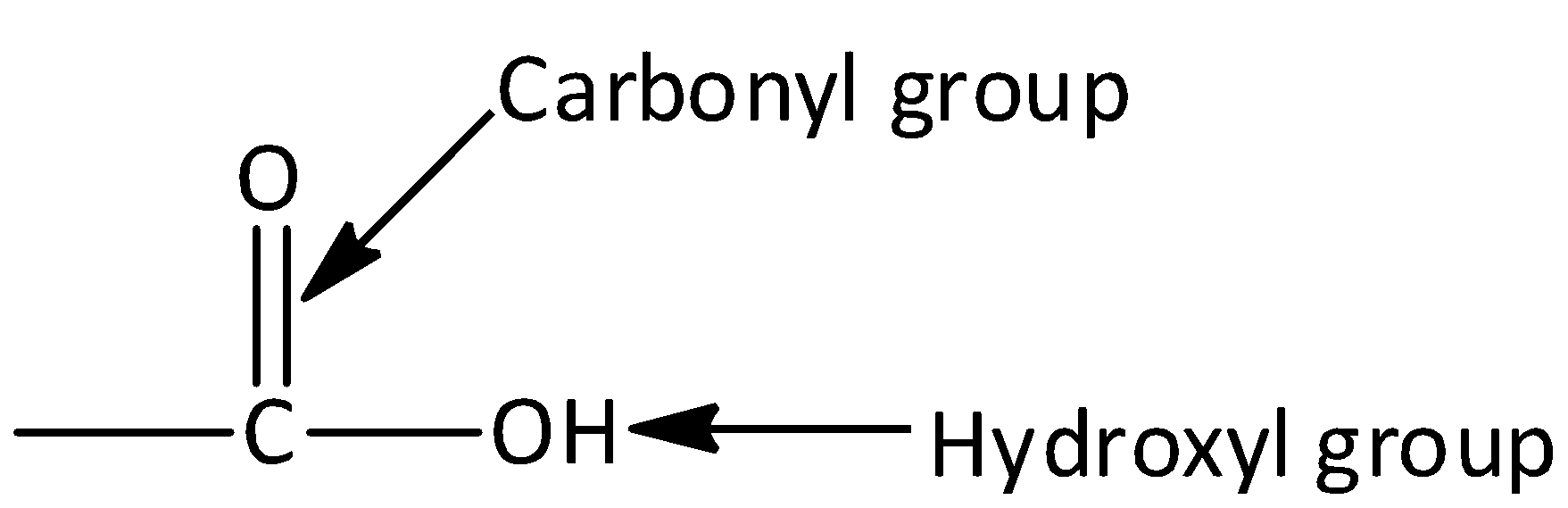
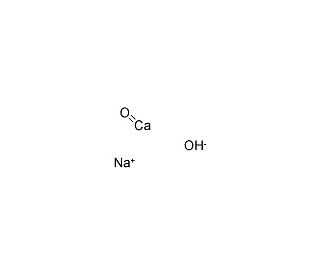
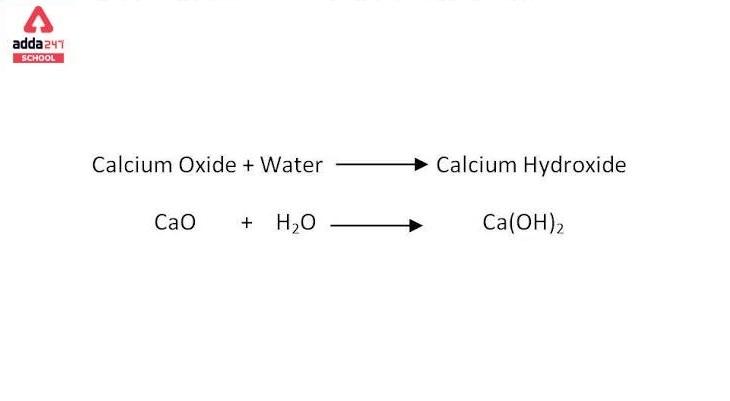
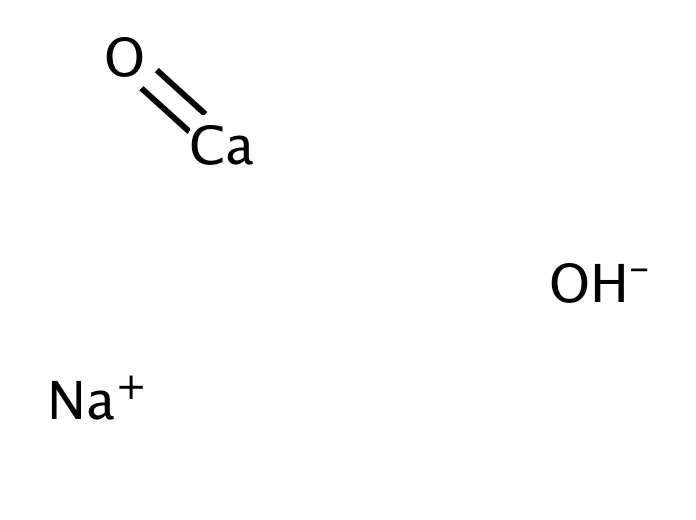
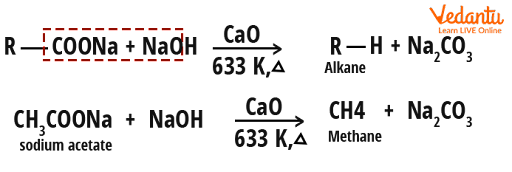explain this line: "quicklime slaked with soda gives sodalime being a basic oxide it combines with acidic oxide at high - Chemistry - The p-Block Elements - 15108261 | Meritnation.com

An organic compound (A) (C5H8O3) on heating with soda lime gives (B) which reacts with HCN to give (C) . The compound (C) reacts with thionyl chloride to produce (D) which on